Soda Ash
Soda ash is the commercial name of sodium carbonate, with the chemical formula Na2CO3, is a multi-purpose chemical with different applications in different industrial sectors. As is clear from its chemical formula, its soda ash is the sodium salt of carbonic acid and is normally a white solid. Until the middle of the 19th century, this chemical was mainly extracted from plant ashes, and in 1791, the French chemist Nicolas Lebance prepared this substance in its present form. Figure (1) shows the structural formula of soda ash.
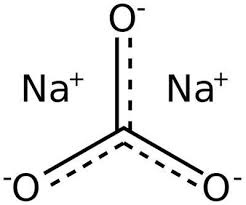
Figure (1): The structural formula of its soda ash
Sodium carbonate production process (Solvay process)
The raw materials required for the production of sodium carbonate by the Solvay method are limestone, common salt and ammonia, which are produced according to the reactions below sodium carbonate.
CaCO3 -> CO2 + CaO
2NH4Cl + CaO + 2NH3 -> CaCl2 + H2O
2NaHCO3 -> Na2CO3 + H2O + CO2
The sodium carbonate produced is divided into two categories in terms of specifications:
- Heavy sodium carbonate
- Light sodium carbonate
The difference between heavy sodium carbonate and light sodium carbonate is only in their density, particle size and application, and they are not chemically different from each other. Heavy sodium carbonate has a bulk density of about 1000 kg / m3 and particles with a size of 300 to 500 microns. Larger granular particles such as this cause the absence of soil dust and impurities and reduce the possibility of particles spreading during transport. The density of light sodium carbonate is about 500 kg / m3 and the particle size of light sodium carbonate is about 100 microns. Heavy soda ash is often used to make glass, while light soda ash is used to make chemicals and detergents.
Soda Ash application
About 45% of the soda ash produced in the world is used in the glass industry. The use of alkalis such as Na2CO3 in glassmaking reduces the glass formation temperature from 1700 to 1500-1450 ° C, which leads to a reduction in energy consumption in this industry. Other uses for soda ash include:
- Oil industry: for deposition of calcium ions in aqueous florets and barium sulfate in low pH florets
- Dyeing: as a bonding agent between dyes and fibers
- Food industry: as a pH regulator and preservative
- Electrolysis: As an electrolyte to increase the rate of water decomposition
- Chemistry Laboratories: As the primary standard in titration reactions

