Review the catalyst industry in Iran (petrochemical catalysts)
Petrochemical complexes use various processes to convert hydrocarbon and natural gas sections into final products. Typically, the processes in petrochemical complexes can be divided into the following two categories:
- Polymeric
- Non-polymeric
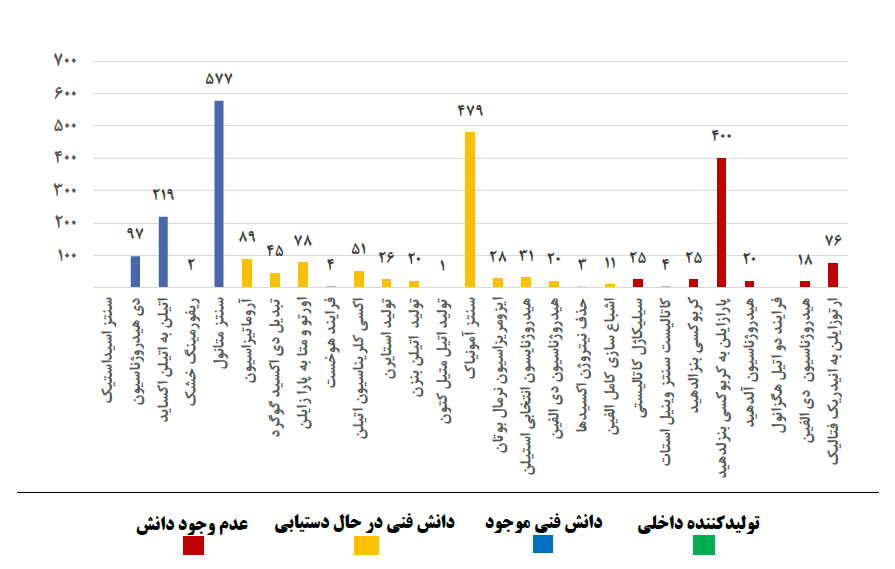
Figures 1 and 2 show the consumption volume and technology status of each of the non-polymer and polymer catalysts in the petrochemical industry, respectively.
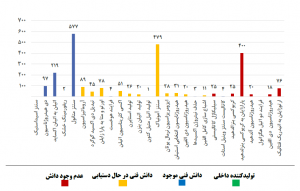
Fig (1): Consumption volume and technology status of each of the non-polymer catalysts

Fig (2): Consumption volume and technology status of each polymer catalyst
As can be seen, unlike refinery catalysts, the catalysts used in the petrochemical industry are very diverse and the volume of demand is lower compared to refinery catalysts. Due to the variety and multiplicity of catalysts used in petrochemical complexes, as shown in Figures (1) and (2), a significant part of the catalysts used in this part of the industry, are imported directly from abroad. Despite the fundamental differences between the two refining and petrochemical industries in terms of raw materials and products, there are similar process units in both industries. Some common processes in refining and petrochemical complexes are:
- Steam Reforming to produce hydrogen and synthesize gas
- Water – Low Temperature (LTSC) and High Temperature (HTSC) gas displacement in synthesis and hydrogen production processes
- Sulfur Recovery (SRU)
- Removal of sulfur (zinc oxide)
Figure (3) shows the consumption volume of each of these catalysts separately. Due to the less complexity of common catalysts and also the low process sensitivities of these catalysts, most of these catalysts are produced and supplied by domestic companies.
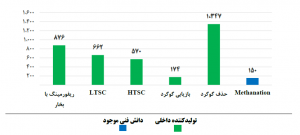
Fig (3): Consumption volume of each of these common catalysts in refining and petrochemical industries