Cyclohexylamine
Cyclohexylamine is an organic compound of the aliphatic amine group, also known as aminocyclohexane and cyclohexane amine. The chemical formula of this colorless liquid is C3H13N. However, like other amines, it may appear colored due to the presence of contaminants. It smells like fish and can be mixed with water and other organic solvents such as alcohols, ethers, ketones and aliphatic and aromatic esters. It was introduced in 1893 but was not used economically in the United States until 1936. But today it is one of the most highly produced chemicals in the chemical industry and has an annual production of over one million in the United States. The structural buffer of this chemical is given in Figure (1).
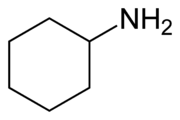
Figure (1): Structural formula of cyclohexylamine
The standard and permissible amount of cyclohexylamine for workers exposed to this substance is 10 ppm in an 8-hour shift. Because it is highly flammable, it can also ignite at ambient temperature, releasing nitrogen oxides during combustion, which can also affect the nervous system. For this reason, it is essential that the product be carefully studied by people who are exposed to the substance before working with MSDS.
Cyclohexylamines, like other amines, are weak bases compared to strong bases such as sodium hydroxide. Although aniline and cyclohexyl amine both have the NH2 group and a hexagonal carbon ring, the difference in the base strength of the two substances can be attributed to their structure. And because of this, the chemical is weaker than aniline.
This material is produced using two methods. These processes include:
- Hydrogenation of aniline in the presence of cobalt or nickel as catalyst:
C6H5NH2 + 3H2 -> C6H11NH2
- Using ammonia alkylation reaction in the presence of cyclohexanol
According to reports published in 2016, Asia, especially China, accounts for about 65% of total global capacity. Due to the increasing demand for nylon, the demand for this material has been growing. After China, Japan and Taiwan are also known as the largest producers of this substance, which accounted for 80% of total global production in 2016.
Applications of cyclohexyl amine include the following:
- Pharmaceutical industry: Production of analgesics
- Agricultural Industries: Production of some herbicides
- Oil and gas industry:
- The compound mediates the synthesis of many organic compounds
- Inhibitor against corrosion and sediment
- Neutralizing raw material
- Water purifiers for boilers
- Petroleum products additive
- Cosmetics industry: as raw materials for the production of some perfumes
- Printing and dyeing industries: as a solvent
- Water and wastewater treatment
- Production of PVC adhesive
